Newborn Screening
Why we need newborn screening for Metachromatic Leukodystrophy
- MLD is a devastating and progressive genetic condition. The majority of children diagnosed with MLD will die before their fifth birthday.
- This no longer needs to be the case. Since 2022, a gene therapy has been approved by NICE and rolled out across the NHS. This treatment is effective if it is administered before irreversible symptoms take effect – this early diagnosis is best achieved through newborn screening.
- Without screening, diagnosis and therefore treatment is a lottery with only 8 out of 40 children diagnosed with MLD since 2022 being treated. Experts predict that another 30 to 40 children will die who do not need to before the government plans to reconsider its decision in 2029/30.
- Parents describe watching their children “lose the ability to walk, talk, see, hear and swallow… to endure epilepsy and dementia” – a level of appalling suffering that is now preventable with early diagnosis.
- These courageous family stories illustrate the stark reality of patients living with untreated MLD versus the transformational power of early treatment intervention:
- Minsters now have the opportunity to save an average of one child per month simply by adding MLD to the UK’s newborn screening programme.
The current position
- The UK National Screening Committee (NSC) has recommended against screening for MLD.
- Experts believe that the process followed to assess MLD screening was procedurally, methodologically, and scientifically flawed.
- The expectation for evidence was impossible to meet, requiring data that can only come from live screening, while refusing to allow screening to begin.
- Data was requested that could not have been collected in an ethical way.
- Peer-reviewed and published evidence was ignored.
- Relevant experts weren’t included in the process.
- The UK is falling behind internationally. The UK screens for just ten conditions at birth and has only added one condition in the last decade, whereas other countries across Europe screen for 30-40 conditions and, on average, have added 19 conditions in just five years.
- The NSC’s broken evidence review process is causing children to die needlessly.
Why we are campaigning for screening to begin now
- Government policy rightly emphasises earlier diagnosis and prevention.
- MLD is a clear case where early diagnosis directly determines outcome and treatment is already available.
- Delay is not neutral – it has real and irreversible consequences.
- The case for screening for MLD is clear-cut and is supported by leading medical and clinical experts both in the UK and internationally.
- This is a moment where ministerial leadership can genuinely make a direct, immediate, and measurable difference.
- Screening for MLD could be introduced quickly if there is the political will to do so, either by adding MLD to the newborn programme or through some form of national pilot or conditional approval.
Further background information
Owing to the rarity of Metachromatic Leukodystrophy (MLD) and other very serious conditions, many children are misdiagnosed or diagnosed at a point where the disease has already progressed too far for cutting-edge treatments such as gene therapy to be of benefit.
ArchAngel is dedicated to ensuring that Metachromatic Leukodystrophy (MLD) is added to the UK’s Newborn Bloodspot Screening (NBS) Programme to guarantee timely diagnosis. The Trust has established an MLD NBS Steering Group of highly specialist scientists and clinicians to carry out the work required and provides the group secretariat. Progress on the application to date is as follows:
In December 2021 the steering group submitted a formal application to the National Screening Committee (NSC).
In April 2022 the UK NSC Secretariat confirmed it would commission an ‘evidence map’ to further examine the case for MLD.
In June 2023 the NSC published an evidence review, stating that “the available evidence on screening test accuracy and cost-effectiveness, though limited, is promising and warrants further review. It found that the volume and type of evidence related to the benefits and harms of treatments in presymptomatic patients with MLD is sufficient to justify a more in-depth review of the evidence”.
June 2023 MLD was placed in the standard the 3-year review cycle. The case will therefore be reviewed in 2026, unless significant new evidence is available prior to that date, in which case the steering group could apply for a rapid review. The steering group is actively working on producing and gathering further evidence to meet the NSC’s requirements.
In February 2024 Wu et al. published very favourable results of a pre-pilot study of the performance of a test developed by Prof. Michael Gelb in the US. ArchAngel is extremely grateful to the high number of UK MLD families who made this vital research possible by authorising use of their MLD affected child’s bloodspot card to provide accurate control material. The next stage of this work is a pilot study, which is currently in planning and seeking necessary permissions.
- In May 2025 the UK NSC opened a 3-month public consultation into proposed NBS for MLD, with guidance that the NSC would NOT support recommendation to approve. The NSC received 501 responses expressing overwhelming support for the approval of screening. We sincerely thank everyone who took the time to express their professional and personal opinions in response to the consultation.
- In January 2026 the NSC also published an announcement regarding the proposed EquipoISE project, essentially is a multi-condition evaluation study, citing MLD as a potential candidate. However, our MLD expert steering group do not believe that this proposal is an appropriate solution for MLD, due to both unacceptable delay and technical incompatibility. We therefore continue to campaign for urgent ministerial intervention.
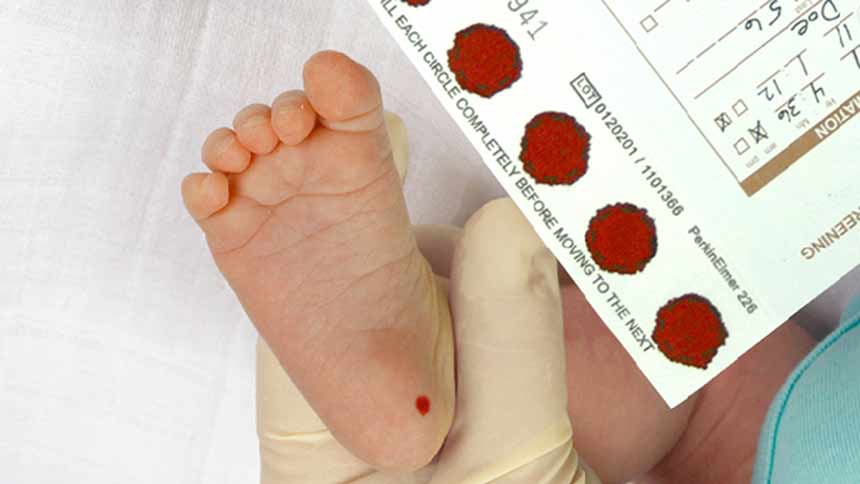
Rare Diseases disproportionally affect children and diagnostic delays result in unnecessary suffering and undue burden on the health service. Newborn screening has the power to dramatically improve children’s lives, by facilitating early access to treatment, maximising the chance for eligibility for treatments and reducing the chance for serious effects of disease prior to treatment.
In the United Kingdom (UK) babies are screened via the heel-prick test (also known as the blood spot or Guthrie test) at 5 or 6 days old. A midwife visits the family home and takes a blood sample by making a small prick on the baby’s heel. The blood sample is then sent to a laboratory, where it is analysed or ‘screened’ for 9 rare disorders. Many other high-income countries screening for a much greater number of conditions. However, the current system for accepting new disorders onto the UK programme remains a very complex and lengthy process. The criteria and standards for screening are set and monitored by the National Screening Committee and Public Health England.
England Rare Disease Action Plan
In February 2022, the DHSC published the England Rare Disease Action Plan containing an important commitment to improving newborn screening, in line with the key policy changes which the Collaborative has been calling for:
You can read the full England Rare Disease Action Plan here:
The Collaborative will continue to work with the DHSC on the detail of the above promises at every opportunity and hope that there will be more positive news to share in the coming months.
In the meantime, exciting developments in Genomic screening has raised many questions about how and when this will fit into the current programme, the attached briefing paper outlines the considerations which we are exploring with experts:
Click here to download NBS Genomics Briefing Paper
In 2020 ArchAngel MLD Trust brought together 13 principal rare disease patient organisations, plus Genetic Alliance UK, to form a new UK NBS Collaborative, representing over 500 rare diseases and thousands of patients. The Collaborative [link] actively campaigns for key policy changes which would allow for the appropriate and efficient expansion of the UK NBS programme, in order to improve the prospects of all future MLD affected patients and many other rare diseases where treatments and tests are available.
The Collaborative was inspired by the important work of Pat Roberts, who has been dedicated to this field for over 20 years through close family experience of another IMD, Krabbe disease. She founded both the Save Babies Through Screening Foundation UK and the Patient Advocates for Newborn Screening Group, and has worked extensively alongside scientists, Public Health England and numerous other key health professionals on the extension of the newborn screening programme in the UK for all inherited metabolic disorders. ArchAngel MLD Trust is incredibly lucky to have Pat as Director of Newborn Screening Project.